Estrogen Receptor Alpha (N-Terminal) CUTANA™ CUT&RUN Antibody
{"url":"https://www.epicypher.com/products/antibodies/cutana-cut-run-antibodies/estrogen-receptor-alpha-n-terminal-cutana-cut-run-antibody","add_this":[{"service":"facebook","annotation":""},{"service":"email","annotation":""},{"service":"print","annotation":""},{"service":"twitter","annotation":""},{"service":"linkedin","annotation":""}],"gtin":null,"id":840,"bulk_discount_rates":[],"can_purchase":true,"meta_description":"Polyclonal anti-Estrogen Receptor Alpha antibody validated for CUT&RUN, IP, IHC. Rigorously tested for reliable and robust performance in CUT&RUN assays.","category":["Chromatin Mapping/CUTANA™ ChIC / CUT&RUN Assays","Antibodies/CUTANA™ CUT&RUN Antibodies","Antibodies/CUTANA™ CUT&RUN Antibodies/CUT&RUN Antibodies - Chromatin-Associated Proteins"],"AddThisServiceButtonMeta":"","main_image":{"data":"https://cdn11.bigcommerce.com/s-y9o92/images/stencil/{:size}/products/840/955/estrogen-receptor-alpha-n-terminal-cutana-cutandrun-antibody__87451.1645734547.jpg?c=2","alt":"Estrogen Receptor Alpha N-Terminal CUTANA CUTandRUN Antibody"},"add_to_wishlist_url":"/wishlist.php?action=add&product_id=840","shipping":{"calculated":true},"num_reviews":0,"weight":"0.00 LBS","custom_fields":[{"id":"711","name":"Pack size","value":"100 µL"}],"sku":"13-2011","description":"<div class=\"product-general-info\">\n <ul class=\"product-general-info__list-left\">\n <li class=\"product-general-info__list-item\">\n <strong>Type: </strong>Polyclonal\n </li>\n <li class=\"product-general-info__list-item\">\n <strong>Host: </strong>Rabbit\n </li>\n <li class=\"product-general-info__list-item\">\n <strong>Applications: </strong>CUT&RUN, IP, IHC\n </li>\n </ul>\n <ul class=\"product-general-info__list-right\">\n <li class=\"product-general-info__list-item\">\n <strong>Reactivity: </strong>Human, Mouse (predicted), Rat\n (predicted)\n </li>\n <li class=\"product-general-info__list-item\">\n <strong>Format: </strong>Antigen affinity-purified\n </li>\n <li class=\"product-general-info__list-item\">\n <strong>Target Size: </strong>66 kDa\n </li>\n </ul>\n</div>\n<div class=\"service_accordion product-droppdown\">\n <div class=\"container\">\n <div id=\"prodAccordion\">\n <div id=\"ProductDescription\" class=\"Block Panel current\">\n <h3 class=\"sub-title1\">Description</h3>\n <div\n class=\"ProductDescriptionContainer product-droppdown__section-description-specific\">\n <p>\n This antibody meets EpiCypher’s \"CUTANA Compatible\" criteria for\n performance in Cleavage Under Targets and Release Using Nuclease\n (CUT&RUN) and/or Cleavage Under Targets and Tagmentation (CUT&Tag)\n approaches to genomic mapping. Every lot of a CUTANA Compatible\n antibody is tested in the indicated approach using EpiCypher\n optimized\n <a href=\"/resources/protocols\"> protocols </a>\n and determined to yield peaks that show a genomic distribution\n pattern consistent with reported function(s) of the target protein.\n Estrogen Receptor Alpha N-terminal (ER alpha N-term) antibody shows\n CUT&RUN peaks in response to estradiol stimulation (<strong\n >Figure 1</strong\n >) that overlap with known estrogen response element (ERE) binding\n motifs (<strong>Figure 2</strong>). Overlap is further observed with\n peaks from an antibody to a different ER alpha epitope (C-term) and\n NCOA3 (SRC3), which co-activates ER-mediated transcription [1]\n (<strong>Figure 2</strong>).\n </p>\n </div>\n </div>\n </div>\n <div id=\"prodAccordion\">\n <div id=\"ProductDescription\" class=\"Block Panel current\">\n <h3 class=\"sub-title1\">Validation Data</h3>\n <div\n class=\"ProductDescriptionContainer product-droppdown__section-description-specific\">\n <p>\n Serum-starved MCF7 cells were treated with estradiol (E2) or vehicle\n control for 45 minutes. CUT&RUN was performed on 500k cells with 0.5\n µg of either ER Alpha (N-Term), ER Alpha (C-Term; EpiCypher\n <a\n href=\"/products/antibodies/cutana-cut-run-antibodies/estrogen-receptor-alpha-c-terminal-cutana-cut-run-antibody\"\n >13-2012</a\n >), NOCA3/SRC3 (EpiCypher\n <a\n href=\"/products/antibodies/cutana-cut-run-antibodies/ncoa3-src3-cutana-cut-run-antibody\"\n >13-2013</a\n >), H3K4me3 positive control (EpiCypher\n <a\n href=\"/products/antibodies/snap-chip-certified-antibodies/histone-h3k4me3-antibody-snap-chip-certified-cutana-cut-run-compatible\"\n >13-0041</a\n >), or IgG negative control (EpiCypher\n <a\n href=\"/products/nucleosomes/snap-cutana-spike-in-controls/cutana-rabbit-igg-cut-run-negative-control-antibody\"\n >13-0042</a\n >) antibodies using the CUTANA™ ChIC/CUT&RUN Kit v2.0 (EpiCypher\n <a\n href=\"/products/epigenetics-reagents-and-assays/cutana-chic-cut-and-run-kit\"\n >14-1048</a\n >). Library preparation was performed with 5 ng of DNA (or the total\n amount recovered if less than 5 ng) using the CUTANA™ CUT&RUN\n Library Prep Kit (EpiCypher\n <a\n href=\"/products/epigenetics-reagents-and-assays/cutana-cut-and-run-library-prep-kit\"\n >14-1001/14-1002</a\n >). Both kit protocols were adapted for high throughput Tecan liquid\n handling. Libraries were run on an Illumina NextSeq2000 with\n paired-end sequencing (2x50 bp). Data were aligned to the hg19\n genome using Bowtie2. Data were filtered to remove duplicates,\n multi-aligned reads, and ENCODE DAC Exclusion List regions.\n </p>\n\n <section class=\"image-picker\">\n <div class=\"image-picker__left\">\n <div\n class=\"image-picker__main-content_active image-picker__main-content\">\n <div class=\"image-picker__header-content\">\n <button class=\"image-picker__left-arrow\">\n <svg\n class=\"image-picker__svg-left\"\n width=\"24\"\n height=\"24\"\n viewBox=\"0 0 24 24\">\n <path\n d=\"M16.67 0l2.83 2.829-9.339 9.175 9.339 9.167-2.83 2.829-12.17-11.996z\" />\n </svg>\n </button>\n <a\n href=\"/content/images/products/antibodies/13_2011_Heatmap.jpeg\"\n target=\"_new\"\n class=\"image-picker__main-image-link\"\n ><img loading=\"lazy\"\n alt=\"13_2011_Heatmap\"\n src=\"/content/images/products/antibodies/13_2011_Heatmap.jpeg\"\n class=\"image-picker__main-image\" />\n <span class=\"image-picker__main-image-caption\"\n >(Click to enlarge)</span\n ></a\n >\n <button class=\"image-picker__right-arrow\">\n <svg\n class=\"image-picker__svg-right\"\n width=\"24\"\n height=\"24\"\n viewBox=\"0 0 24 24\">\n <path\n d=\"M7.33 24l-2.83-2.829 9.339-9.175-9.339-9.167 2.83-2.829 12.17 11.996z\" />\n </svg>\n </button>\n </div>\n <p>\n <span class=\"image-picker__span-content\"\n ><strong>Figure 1: ER alpha N-term peaks in CUT&RUN </strong\n ><br />\n CUT&RUN was performed as described above. Heatmaps show ER\n alpha N-term peaks relative to IgG negative control, H3K4me3\n positive control, ER alpha C-term, and SRC3 antibodies in\n aligned rows ranked by intensity (top to bottom) and colored\n such that red indicates high localized enrichment and blue\n denotes background signal.\n </span>\n </p>\n </div>\n <div class=\"image-picker__main-content\">\n <div class=\"image-picker__header-content\">\n <button class=\"image-picker__left-arrow\">\n <svg\n class=\"image-picker__svg-left\"\n width=\"24\"\n height=\"24\"\n viewBox=\"0 0 24 24\">\n <path\n d=\"M16.67 0l2.83 2.829-9.339 9.175 9.339 9.167-2.83 2.829-12.17-11.996z\" />\n </svg>\n </button>\n <a\n href=\"/content/images/products/antibodies/13_2011_Peaks.jpeg\"\n target=\"_new\"\n class=\"image-picker__main-image-link\"\n ><img loading=\"lazy\"\n alt=\"13_2011_Peaks\"\n src=\"/content/images/products/antibodies/13_2011_Peaks.jpeg\"\n class=\"image-picker__main-image\" />\n <span class=\"image-picker__main-image-caption\"\n >(Click to enlarge)</span\n ></a\n >\n <button class=\"image-picker__right-arrow\">\n <svg\n class=\"image-picker__svg-right\"\n width=\"24\"\n height=\"24\"\n viewBox=\"0 0 24 24\">\n <path\n d=\"M7.33 24l-2.83-2.829 9.339-9.175-9.339-9.167 2.83-2.829 12.17 11.996z\" />\n </svg>\n </button>\n </div>\n <p>\n <span class=\"image-picker__span-content\"\n ><strong\n >Figure 2: ER alpha N-term peak analysis in\n CUT&RUN</strong\n ><br />\n Peaks from the E2-treated samples in Figure 1 were called\n using MACS2. (<strong>A</strong>) The number of ER alpha\n N-term peaks which fall into distinct classes of\n functionally annotated genomic regions is plotted.\n (<strong>B</strong>) Homer analysis determined that the ERE\n consensus motif, represented as a sequence logo position\n weight matrix, was enriched under ER alpha N-term peaks.\n (<strong>C</strong>) The number of ER alpha N-term peaks\n containing consensus motifs from panel B is shown by Venn\n Diagram. (<strong>D</strong>) The number of ER alpha N-term\n peaks that overlap with ER alpha C-term and SRC3 antibodies\n are represented by Venn Diagram.\n </span>\n </p>\n </div>\n <div class=\"image-picker__main-content\">\n <div class=\"image-picker__header-content\">\n <button class=\"image-picker__left-arrow\">\n <svg\n class=\"image-picker__svg-left\"\n width=\"24\"\n height=\"24\"\n viewBox=\"0 0 24 24\">\n <path\n d=\"M16.67 0l2.83 2.829-9.339 9.175 9.339 9.167-2.83 2.829-12.17-11.996z\" />\n </svg>\n </button>\n <a\n href=\"/content/images/products/antibodies/13_2011_IP.jpeg\"\n target=\"_new\"\n class=\"image-picker__main-image-link\"\n ><img loading=\"lazy\"\n alt=\"13_2011_IP\"\n src=\"/content/images/products/antibodies/13_2011_IP.jpeg\"\n class=\"image-picker__main-image\" />\n <span class=\"image-picker__main-image-caption\"\n >(Click to enlarge)</span\n ></a\n >\n <button class=\"image-picker__right-arrow\">\n <svg\n class=\"image-picker__svg-right\"\n width=\"24\"\n height=\"24\"\n viewBox=\"0 0 24 24\">\n <path\n d=\"M7.33 24l-2.83-2.829 9.339-9.175-9.339-9.167 2.83-2.829 12.17 11.996z\" />\n </svg>\n </button>\n </div>\n <p>\n <span class=\"image-picker__span-content\"\n ><strong>Figure 3: Immunoprecipitation data</strong><br />\n EpiCypher ER alpha N-term antibody (3 µg) was used to\n immunoprecipitate whole cell lysates isolated from MCF7\n cells (1.0 mg per IP). A negative control IgG antibody and\n positive control antibody to different ER alpha epitopes\n (EpiCypher\n <a\n href=\"/products/antibodies/cutana-cut-run-antibodies/estrogen-receptor-alpha-c-terminal-cutana-cut-run-antibody\"\n >13-2012</a\n >\n and Bethyl Laboratories) were also used to demonstrate\n specificity of the IP. Immunoprecipitates were loaded onto a\n 4-8% SDS-PAGE gel (25% of IP loaded) and probed via western\n blot with EpiCypher\n <a\n href=\"/products/antibodies/cutana-cut-run-antibodies/estrogen-receptor-alpha-c-terminal-cutana-cut-run-antibody\"\n >13-2012</a\n >\n ER alpha C-term antibody (0.1 µg/mL).\n </span>\n </p>\n </div>\n <div class=\"image-picker__main-content\">\n <div class=\"image-picker__header-content\">\n <button class=\"image-picker__left-arrow\">\n <svg\n class=\"image-picker__svg-left\"\n width=\"24\"\n height=\"24\"\n viewBox=\"0 0 24 24\">\n <path\n d=\"M16.67 0l2.83 2.829-9.339 9.175 9.339 9.167-2.83 2.829-12.17-11.996z\" />\n </svg>\n </button>\n <a\n href=\"/content/images/products/antibodies/13_2011_IHC.jpeg\"\n target=\"_new\"\n class=\"image-picker__main-image-link\"\n ><img loading=\"lazy\"\n alt=\"13_2011_IHC\"\n src=\"/content/images/products/antibodies/13_2011_IHC.jpeg\"\n class=\"image-picker__main-image\" />\n <span class=\"image-picker__main-image-caption\"\n >(Click to enlarge)</span\n ></a\n >\n <button class=\"image-picker__right-arrow\">\n <svg\n class=\"image-picker__svg-right\"\n width=\"24\"\n height=\"24\"\n viewBox=\"0 0 24 24\">\n <path\n d=\"M7.33 24l-2.83-2.829 9.339-9.175-9.339-9.167 2.83-2.829 12.17 11.996z\" />\n </svg>\n </button>\n </div>\n <p>\n <span class=\"image-picker__span-content\"\n ><strong>Figure 4: Immunohistochemistry data</strong><br />\n FFPE section of human breast examined using ER alpha N-term\n antibody (1:2,500 dilution, 0.4 µg/mL).\n </span>\n </p>\n </div>\n </div>\n <aside class=\"image-picker__right\">\n <div class=\"image-picker__gallery\">\n <img loading=\"lazy\"\n alt=\"13_2011_Heatmap\"\n src=\"/content/images/products/antibodies/13_2011_Heatmap.jpeg\"\n width=\"200\"\n class=\"image-picker__side-image image-picker__side-image_active\"\n role=\"button\" />\n <img loading=\"lazy\"\n alt=\"13_2011_Peaks\"\n src=\"/content/images/products/antibodies/13_2011_Peaks.jpeg\"\n class=\"image-picker__side-image\"\n role=\"button\" />\n <img loading=\"lazy\"\n alt=\"13_2011_IP\"\n src=\"/content/images/products/antibodies/13_2011_IP.jpeg\"\n class=\"image-picker__side-image\"\n role=\"button\" />\n <img loading=\"lazy\"\n alt=\"13_2011_IHC\"\n src=\"/content/images/products/antibodies/13_2011_IHC.jpeg\"\n class=\"image-picker__side-image\"\n role=\"button\" />\n </div>\n </aside>\n </section>\n </div>\n </div>\n </div>\n <div id=\"prodAccordion\">\n <div id=\"ProductDescription\" class=\"Block Panel\">\n <h3 class=\"sub-title1\">Technical Information</h3>\n <div\n class=\"ProductDescriptionContainer product-droppdown__section-description\">\n <div class=\"product-tech-info\">\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>Immunogen</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">\n Between amino acids 1 and 50\n </div>\n </div>\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>Storage</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">\n Stable for 1 year at 4°C from date of receipt\n </div>\n </div>\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>Formulation</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">\n Antigen affinity-purified antibody in Tris-citrate/phosphate\n buffer pH 7-8, 0.09% sodium azide\n </div>\n </div>\n </div>\n </div>\n </div>\n </div>\n <div id=\"prodAccordion\">\n <div id=\"ProductDescription\" class=\"Block Panel\">\n <h3 class=\"sub-title1\">Recommended Dilution</h3>\n <div\n class=\"ProductDescriptionContainer product-droppdown__section-description\">\n <div class=\"product-tech-info\">\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>CUT&RUN:</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">\n 0.5 µg per reaction\n </div>\n </div>\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>Immunoprecipitation (IP):</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">\n 3 - 5 µg/mg lysate\n </div>\n </div>\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>Immunohistochemistry (IHC):</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">\n 1:1,000 - 1:5,000. Epitope retrieval with citrate buffer pH 9.0\n is recommended for FFPE tissue sections\n </div>\n </div>\n </div>\n </div>\n </div>\n </div>\n <div id=\"prodAccordion\">\n <div id=\"ProductDescription\" class=\"Block Panel\">\n <h3 class=\"sub-title1\">Gene & Protein Information</h3>\n <div\n class=\"ProductDescriptionContainer product-droppdown__section-description\">\n <div class=\"product-tech-info\">\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>UniProt ID</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">P03372</div>\n </div>\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>Gene Name</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">ESR1</div>\n </div>\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>Protein Name</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">\n Estrogen receptor\n </div>\n </div>\n <div class=\"product-tech-info__line-item\">\n <div class=\"product-tech-info__line-item-left\">\n <b>Alternate Names</b>\n </div>\n <div class=\"product-tech-info__line-item-right\">\n ER, ER-alpha, Estradiol receptor, Nuclear receptor subfamily 3\n group A member 1, ESR, NR3A1\n </div>\n </div>\n </div>\n </div>\n </div>\n </div>\n <div id=\"prodAccordion\">\n <div id=\"ProductDescription\" class=\"Block Panel\">\n <h3 class=\"sub-title1\">References</h3>\n <div\n class=\"ProductDescriptionContainer product-droppdown__section-description\">\n <strong>Background References:</strong>\n <br />\n [1] Wagner et al. <em>BMC Cancer</em> (2013). PMID:\n <a\n href=\"https://pubmed.ncbi.nlm.nih.gov/24304549/\"\n title=\"NCOA3 is a selective co-activator of estrogen receptor α-mediated transactivation of PLAC1 in MCF-7 breast cancer cells\"\n target=\"new\">\n 24304549</a\n ><br />\n </div>\n </div>\n </div>\n <div id=\"prodAccordion\">\n <div id=\"ProductDescription\" class=\"Block Panel\">\n <h3 class=\"sub-title1\">Documents & Resources</h3>\n <div\n class=\"ProductDescriptionContainer product-droppdown__section-description\">\n <div class=\"product-documents\">\n <a\n href=\"/content/documents/tds/13-2011.pdf\"\n target=\"_new\"\n class=\"product-documents__link\">\n <svg\n version=\"1.1\"\n id=\"Layer_1\"\n xmlns=\"http://www.w3.org/2000/svg\"\n xmlns:xlink=\"http://www.w3.org/1999/xlink\"\n x=\"0px\"\n y=\"0px\"\n viewBox=\"0 0 228 240\"\n style=\"enable-background: new 0 0 228 240\"\n xml:space=\"preserve\"\n class=\"product-documents__icon\">\n <g>\n <path\n class=\"product-documents__svg-pdf\"\n d=\"M191.92,68.77l-47.69-47.69c-1.33-1.33-3.12-2.08-5.01-2.08H45.09C41.17,19,38,22.17,38,26.09v184.36\n c0,3.92,3.17,7.09,7.09,7.09h141.82c3.92,0,7.09-3.17,7.09-7.09V73.8C194,71.92,193.25,70.1,191.92,68.77z M177.65,77.06h-41.7\n v-41.7L177.65,77.06z M178.05,201.59H53.95V34.95h66.92v47.86c0,5.14,4.17,9.31,9.31,9.31h47.86V201.59z\" />\n </g>\n <rect\n x=\"20\"\n y=\"112\"\n class=\"product-documents__svg-background\"\n width=\"146\"\n height=\"76\" />\n <g>\n <path\n class=\"product-documents__svg-pdf\"\n d=\"M23.83,125.68h22.36c5.29,0,9.41,1.33,12.35,4c2.94,2.67,4.42,6.39,4.42,11.18c0,4.78-1.47,8.51-4.42,11.18\n c-2.94,2.67-7.06,4-12.35,4H34.59v18.29H23.83V125.68z M44.81,147.9c5.38,0,8.07-2.32,8.07-6.97c0-2.39-0.67-4.16-2-5.31\n c-1.33-1.15-3.36-1.73-6.07-1.73H34.59v14.01H44.81z\" />\n <path\n class=\"product-documents__svg-pdf\"\n d=\"M69.92,125.68h18.91c5.29,0,9.84,0.97,13.66,2.9c3.82,1.93,6.74,4.72,8.76,8.35\n c2.02,3.63,3.04,7.98,3.04,13.04c0,5.06-1,9.42-3,13.08c-2,3.66-4.91,6.45-8.73,8.38c-3.82,1.93-8.4,2.9-13.73,2.9H69.92V125.68z\n M88.07,165.63c10.35,0,15.52-5.22,15.52-15.66c0-10.4-5.17-15.59-15.52-15.59h-7.38v31.26H88.07z\" />\n <path\n class=\"product-documents__svg-pdf\"\n d=\"M122.57,125.68h32.84v8.49h-22.22v11.18h20.84v8.49h-20.84v20.49h-10.63V125.68z\" />\n </g>\n </svg>\n <span class=\"product-documents__info\">Technical Datasheet</span>\n </a>\n </div>\n </div>\n </div>\n </div>\n <div id=\"prodAccordion\">\n <div id=\"ProductDescription\" class=\"Block Panel\">\n <h3 class=\"sub-title1\">Additional Info</h3>\n <div\n class=\"ProductDescriptionContainer product-droppdown__section-description\">\n <p>\n This product is provided for commercial sale under license from\n Bethyl Laboratories, Inc.\n </p>\n </div>\n </div>\n </div>\n </div>\n</div>\n","tags":[],"warranty":"","price":{"without_tax":{"formatted":"$525.00","value":525,"currency":"USD"},"tax_label":"Sales Tax"},"detail_messages":"","availability":"","page_title":"Estrogen Receptor Alpha (N-Terminal) Antibody | CUTANA™ CUT&RUN Compatible","cart_url":"https://www.epicypher.com/cart.php","max_purchase_quantity":0,"mpn":null,"upc":null,"options":[],"related_products":[{"id":889,"sku":null,"name":"CUTANA™ CUT&RUN Library Prep Kit","url":"https://www.epicypher.com/products/epigenetics-kits-and-reagents/cutana-cut-run-library-prep-kit","availability":"","rating":null,"brand":{"name":null},"category":["Chromatin Mapping","Chromatin Mapping/CUTANA™ ChIC / CUT&RUN Assays"],"summary":"\n \n \n \n \n \n \n \n ","image":{"data":"https://cdn11.bigcommerce.com/s-y9o92/images/stencil/{:size}/products/889/997/Epicypher_CUTANA_CUTRUN_LibraryPrep_Box__64431.1646406521.png?c=2","alt":"CUTANA™ CUT&RUN Library Prep Kit"},"images":[{"data":"https://cdn11.bigcommerce.com/s-y9o92/images/stencil/{:size}/products/889/997/Epicypher_CUTANA_CUTRUN_LibraryPrep_Box__64431.1646406521.png?c=2","alt":"CUTANA™ CUT&RUN Library Prep Kit"}],"date_added":"31st Jan 2022","pre_order":false,"show_cart_action":true,"has_options":true,"stock_level":null,"low_stock_level":null,"qty_in_cart":0,"custom_fields":[{"id":983,"name":"Pack Size","value":"48 Reactions"}],"num_reviews":null,"weight":{"formatted":"0.01 LBS","value":0.01},"demo":false,"price":{"without_tax":{"currency":"USD","formatted":"$1,525.00","value":1525},"tax_label":"Sales Tax"},"add_to_wishlist_url":"/wishlist.php?action=add&product_id=889"},{"id":694,"sku":null,"name":"CUTANA™ pAG-MNase for ChIC/CUT&RUN Workflows","url":"https://www.epicypher.com/products/epigenetics-reagents-and-assays/cutana-pag-mnase-for-chic-cut-and-run-workflows","availability":"","rating":null,"brand":{"name":null},"category":["Chromatin Mapping","Chromatin Mapping/CUTANA™ ChIC / CUT&RUN Assays"],"summary":"\n \n \n Type: Nuclease\n \n \n Mol Wgt: 43.7 kDa\n \n \n \n ","image":{"data":"https://cdn11.bigcommerce.com/s-y9o92/images/stencil/{:size}/products/694/689/Screen_Shot_2020-02-12_at_11.01.55_AM__17144.1581530752.png?c=2","alt":"CUTANA™ pAG-MNase for ChIC/CUT&RUN Workflows"},"images":[{"data":"https://cdn11.bigcommerce.com/s-y9o92/images/stencil/{:size}/products/694/689/Screen_Shot_2020-02-12_at_11.01.55_AM__17144.1581530752.png?c=2","alt":"CUTANA™ pAG-MNase for ChIC/CUT&RUN Workflows"}],"date_added":"12th Aug 2019","pre_order":false,"show_cart_action":true,"has_options":true,"stock_level":null,"low_stock_level":null,"qty_in_cart":0,"custom_fields":[{"id":1174,"name":"Internal Comment","value":"Excess in bottom of Venom"},{"id":1175,"name":"Internal Comment","value":"Bulk in Psylocke"}],"num_reviews":null,"weight":{"formatted":"0.01 LBS","value":0.01},"demo":false,"price":{"without_tax":{"currency":"USD","formatted":"$335.00","value":335},"price_range":{"min":{"without_tax":{"currency":"USD","formatted":"$335.00","value":335},"tax_label":"Sales Tax"},"max":{"without_tax":{"currency":"USD","formatted":"$1,295.00","value":1295},"tax_label":"Sales Tax"}},"tax_label":"Sales Tax"},"add_to_wishlist_url":"/wishlist.php?action=add&product_id=694"},{"id":758,"sku":"13-0042","name":"CUTANA™ IgG Negative Control Antibody for CUT&RUN and CUT&Tag","url":"https://www.epicypher.com/products/nucleosomes/snap-cutana-spike-in-controls/cutana-igg-negative-control-antibody-for-cut-run-and-cut-tag","availability":"","rating":null,"brand":{"name":null},"category":["Nucleosomes/SNAP-CUTANA™ Spike-in Controls","Antibodies/CUTANA™ CUT&RUN Antibodies","Chromatin Mapping","Chromatin Mapping/CUTANA™ ChIC / CUT&RUN Assays","Chromatin Mapping/CUTANA™ CUT&Tag Assays"],"summary":"\n \n \n Type: Polyclonal\n \n \n Host: Rabbit\n \n \n Applications: CUT&R","image":{"data":"https://cdn11.bigcommerce.com/s-y9o92/images/stencil/{:size}/products/758/739/1579731877.1280.1280__61771.1592491196.png?c=2","alt":"CUTANA™ IgG Negative Control Antibody for CUT&RUN and CUT&Tag"},"images":[{"data":"https://cdn11.bigcommerce.com/s-y9o92/images/stencil/{:size}/products/758/739/1579731877.1280.1280__61771.1592491196.png?c=2","alt":"CUTANA™ IgG Negative Control Antibody for CUT&RUN and CUT&Tag"}],"date_added":"17th Jun 2020","pre_order":false,"show_cart_action":true,"has_options":false,"stock_level":null,"low_stock_level":null,"qty_in_cart":0,"custom_fields":[{"id":1148,"name":"Pack Size","value":"100 µg"},{"id":1149,"name":"Internal Comment","value":"bulk stocks from Thermo in cold room"}],"num_reviews":null,"weight":{"formatted":"0.01 LBS","value":0.01},"demo":false,"add_to_cart_url":"https://www.epicypher.com/cart.php?action=add&product_id=758","price":{"without_tax":{"currency":"USD","formatted":"$105.00","value":105},"tax_label":"Sales Tax"},"add_to_wishlist_url":"/wishlist.php?action=add&product_id=758"}],"shipping_messages":[],"rating":0,"meta_keywords":"estrogen receptor alpha antibody, estrogen receptor antibody, era antibody, erα antibody, CUT&RUN antibody","show_quantity_input":1,"title":"Estrogen Receptor Alpha (N-Terminal) CUTANA™ CUT&RUN Antibody","gift_wrapping_available":false,"min_purchase_quantity":0,"customizations":[],"images":[{"data":"https://cdn11.bigcommerce.com/s-y9o92/images/stencil/{:size}/products/840/955/estrogen-receptor-alpha-n-terminal-cutana-cutandrun-antibody__87451.1645734547.jpg?c=2","alt":"Estrogen Receptor Alpha N-Terminal CUTANA CUTandRUN Antibody"}]} Pack size: 100 µL
- Type: Polyclonal
- Host: Rabbit
- Applications: CUT&RUN, IP, IHC
- Reactivity: Human, Mouse (predicted), Rat (predicted)
- Format: Antigen affinity-purified
- Target Size: 66 kDa
Description
This antibody meets EpiCypher’s "CUTANA Compatible" criteria for performance in Cleavage Under Targets and Release Using Nuclease (CUT&RUN) and/or Cleavage Under Targets and Tagmentation (CUT&Tag) approaches to genomic mapping. Every lot of a CUTANA Compatible antibody is tested in the indicated approach using EpiCypher optimized protocols and determined to yield peaks that show a genomic distribution pattern consistent with reported function(s) of the target protein. Estrogen Receptor Alpha N-terminal (ER alpha N-term) antibody shows CUT&RUN peaks in response to estradiol stimulation (Figure 1) that overlap with known estrogen response element (ERE) binding motifs (Figure 2). Overlap is further observed with peaks from an antibody to a different ER alpha epitope (C-term) and NCOA3 (SRC3), which co-activates ER-mediated transcription [1] (Figure 2).
Validation Data
Serum-starved MCF7 cells were treated with estradiol (E2) or vehicle control for 45 minutes. CUT&RUN was performed on 500k cells with 0.5 µg of either ER Alpha (N-Term), ER Alpha (C-Term; EpiCypher 13-2012), NOCA3/SRC3 (EpiCypher 13-2013), H3K4me3 positive control (EpiCypher 13-0041), or IgG negative control (EpiCypher 13-0042) antibodies using the CUTANA™ ChIC/CUT&RUN Kit v2.0 (EpiCypher 14-1048). Library preparation was performed with 5 ng of DNA (or the total amount recovered if less than 5 ng) using the CUTANA™ CUT&RUN Library Prep Kit (EpiCypher 14-1001/14-1002). Both kit protocols were adapted for high throughput Tecan liquid handling. Libraries were run on an Illumina NextSeq2000 with paired-end sequencing (2x50 bp). Data were aligned to the hg19 genome using Bowtie2. Data were filtered to remove duplicates, multi-aligned reads, and ENCODE DAC Exclusion List regions.
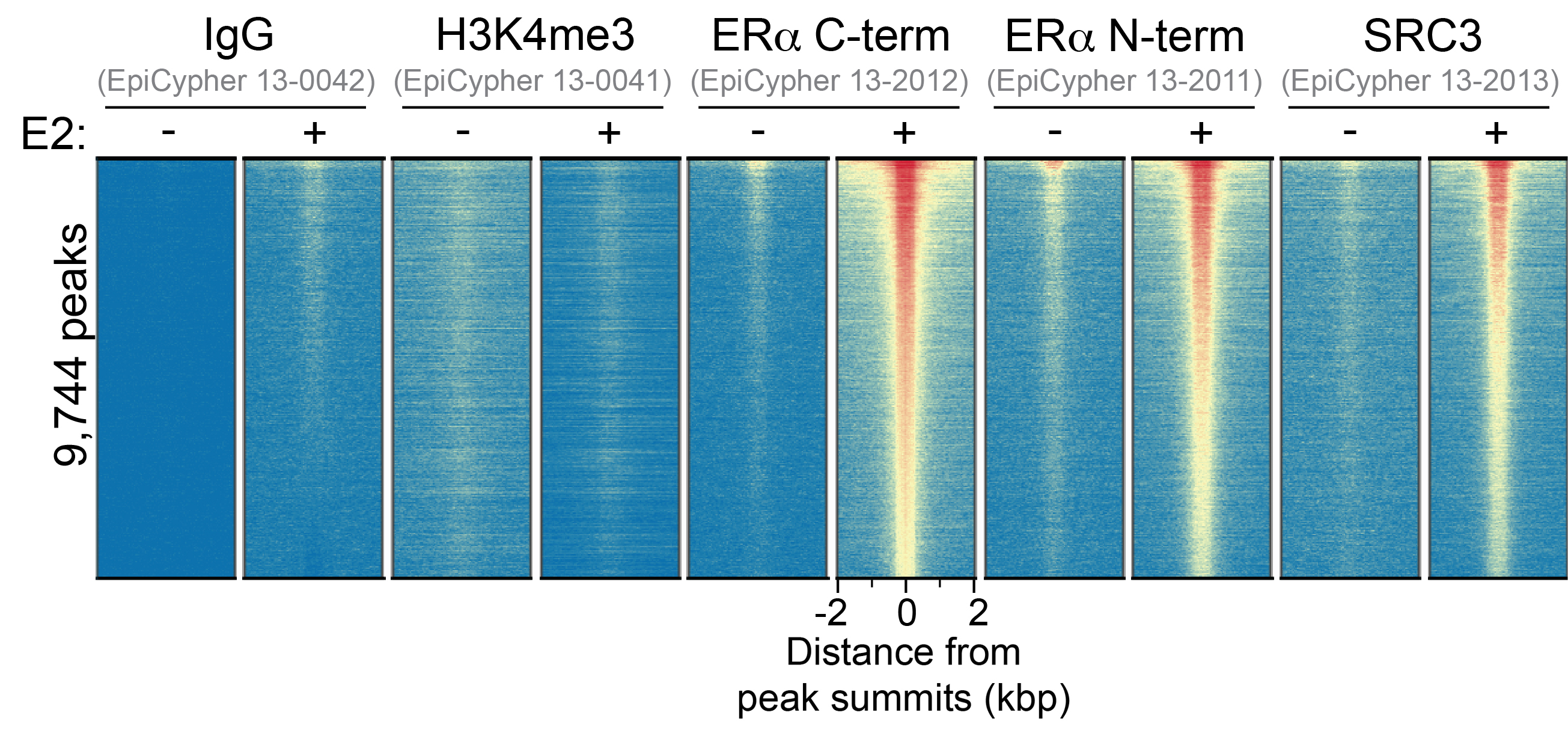
Figure 1: ER alpha N-term peaks in CUT&RUN
CUT&RUN was performed as described above. Heatmaps show ER
alpha N-term peaks relative to IgG negative control, H3K4me3
positive control, ER alpha C-term, and SRC3 antibodies in
aligned rows ranked by intensity (top to bottom) and colored
such that red indicates high localized enrichment and blue
denotes background signal.
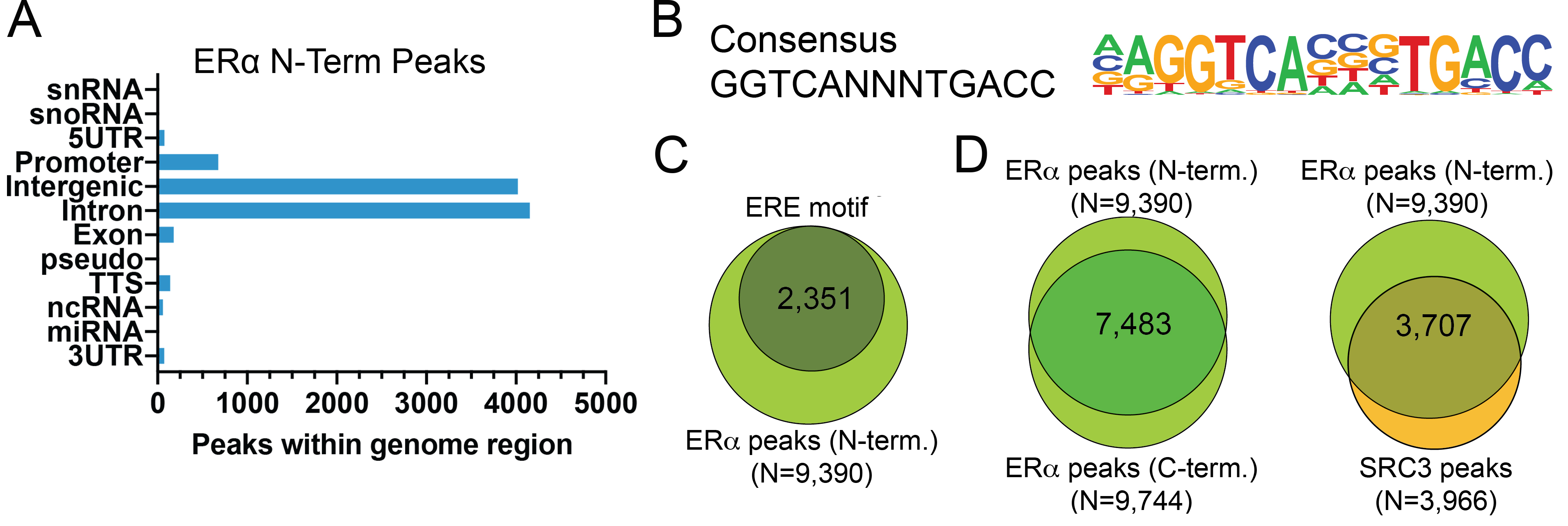
Figure 2: ER alpha N-term peak analysis in
CUT&RUN
Peaks from the E2-treated samples in Figure 1 were called
using MACS2. (A) The number of ER alpha
N-term peaks which fall into distinct classes of
functionally annotated genomic regions is plotted.
(B) Homer analysis determined that the ERE
consensus motif, represented as a sequence logo position
weight matrix, was enriched under ER alpha N-term peaks.
(C) The number of ER alpha N-term peaks
containing consensus motifs from panel B is shown by Venn
Diagram. (D) The number of ER alpha N-term
peaks that overlap with ER alpha C-term and SRC3 antibodies
are represented by Venn Diagram.
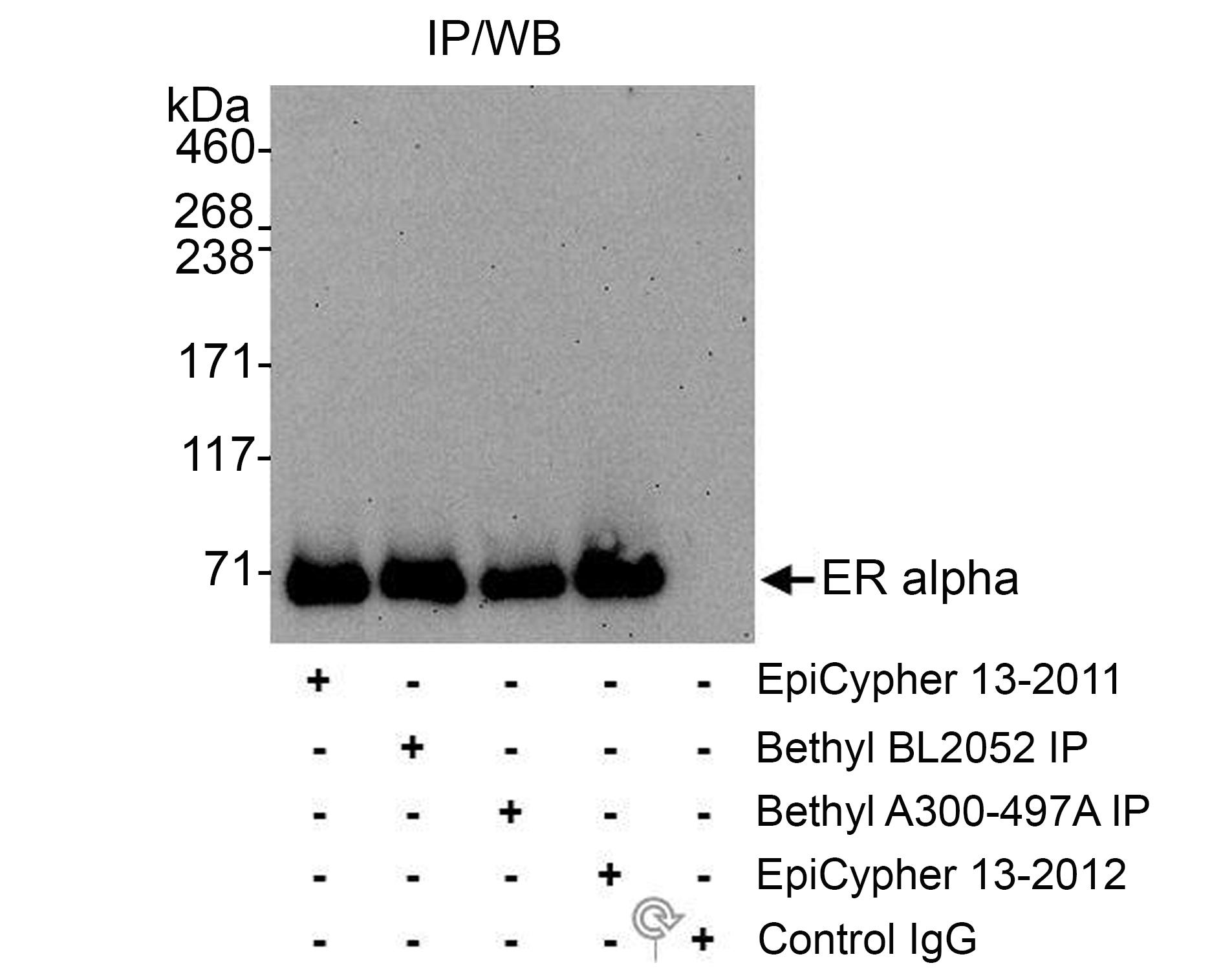
Figure 3: Immunoprecipitation data
EpiCypher ER alpha N-term antibody (3 µg) was used to
immunoprecipitate whole cell lysates isolated from MCF7
cells (1.0 mg per IP). A negative control IgG antibody and
positive control antibody to different ER alpha epitopes
(EpiCypher
13-2012
and Bethyl Laboratories) were also used to demonstrate
specificity of the IP. Immunoprecipitates were loaded onto a
4-8% SDS-PAGE gel (25% of IP loaded) and probed via western
blot with EpiCypher
13-2012
ER alpha C-term antibody (0.1 µg/mL).
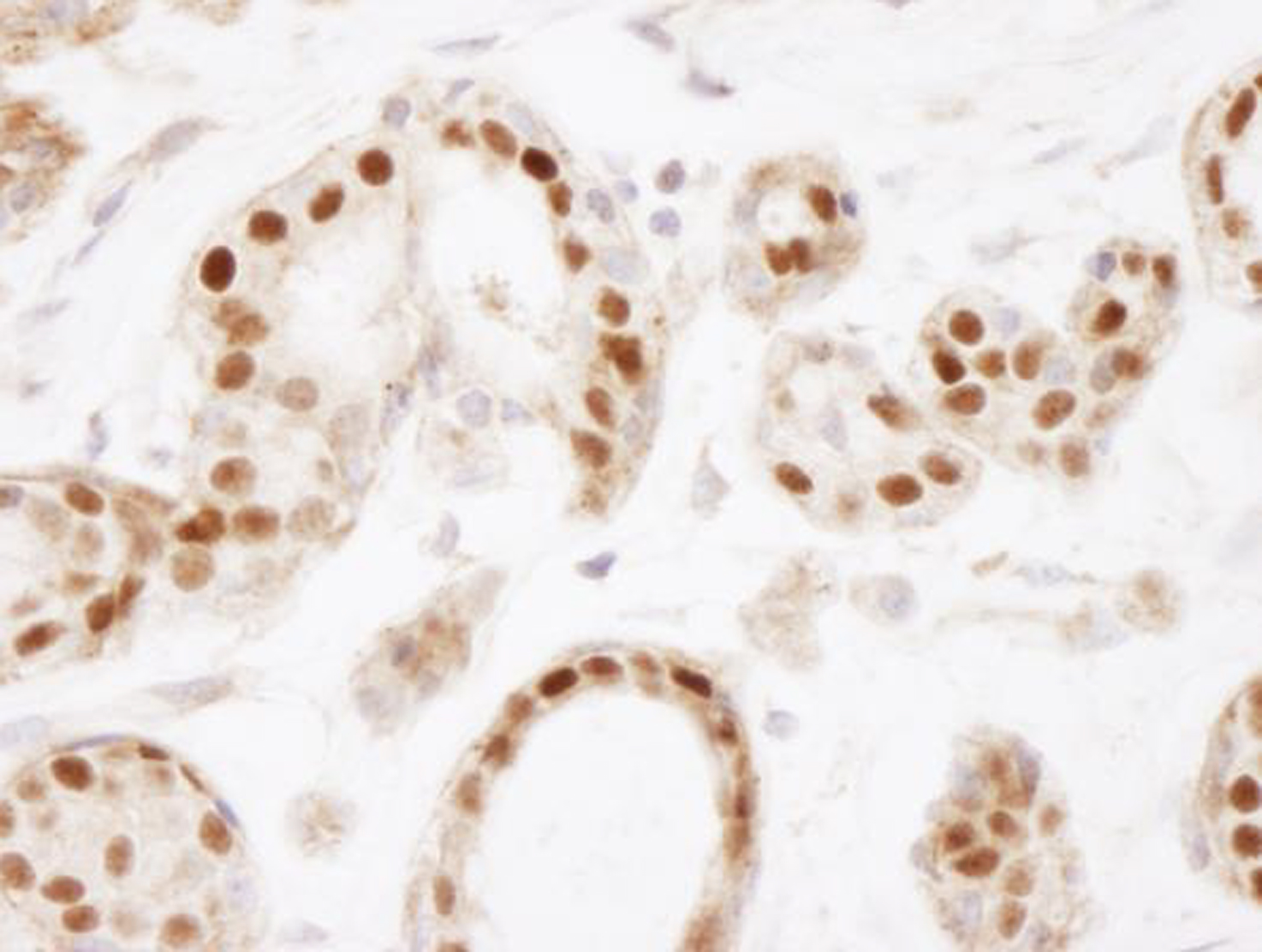
Figure 4: Immunohistochemistry data
FFPE section of human breast examined using ER alpha N-term
antibody (1:2,500 dilution, 0.4 µg/mL).
Technical Information
Recommended Dilution
Gene & Protein Information
References
Documents & Resources
Additional Info
This product is provided for commercial sale under license from Bethyl Laboratories, Inc.