Captify™ Assay Services for Novel Chromatin Interactions
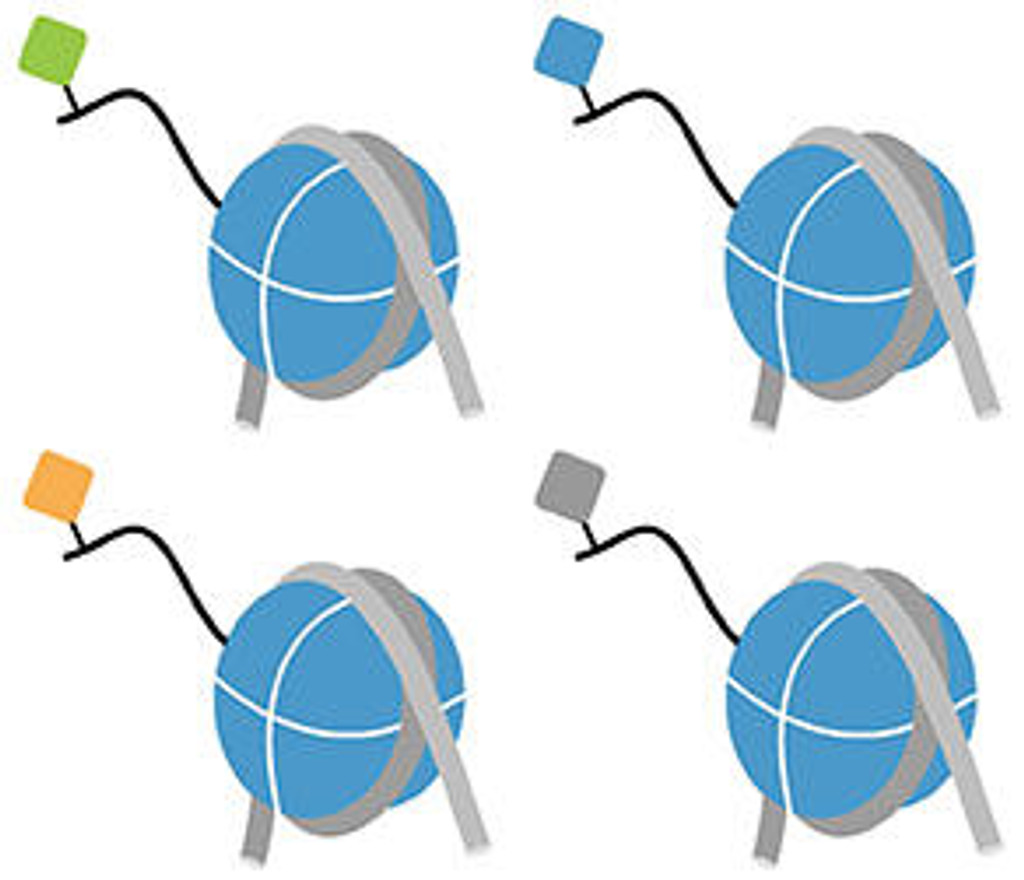
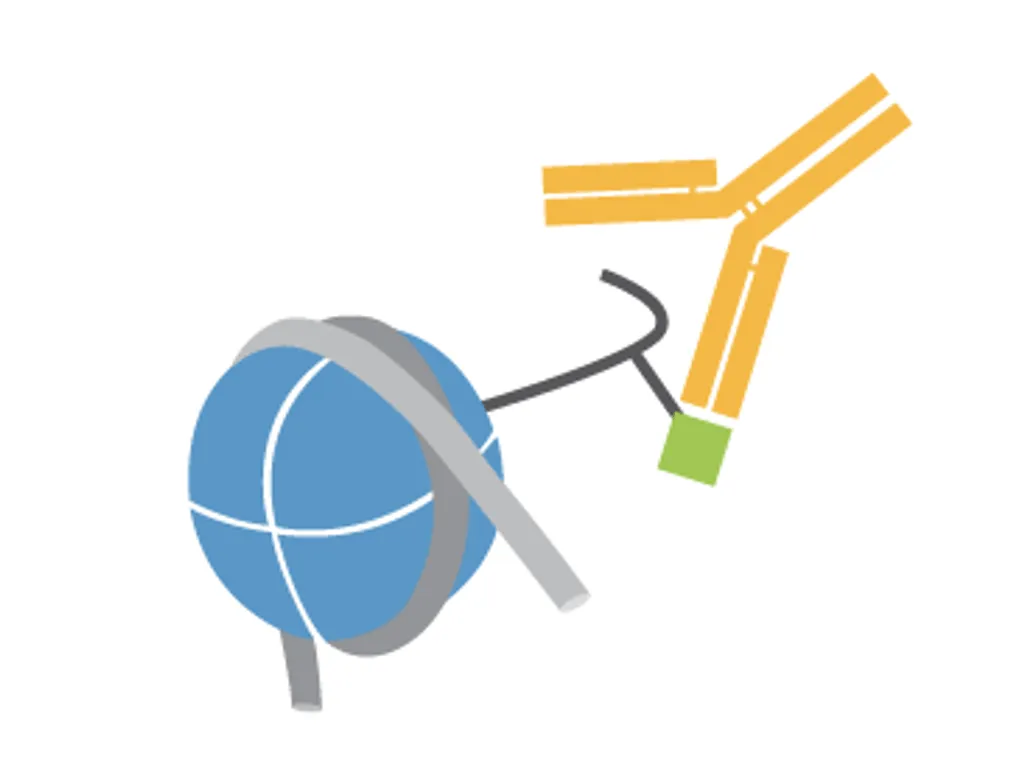
Captify™ assay services reveal the true binding specificity of chromatin readers, enzymes, antibodies, and more. This platform combines EpiCypher’s comprehensive library of recombinant nucleosomes with AlphaScreen® technology, enabling unprecedented throughput and sensitivity. Compared to traditional histone peptide arrays, Captify also has improved physiological accuracy, requires less protein, and is compatible with full length proteins and domains.
Have Questions?
We’re here to help. Click below and a member of our team will get back to you shortly!

Additional Information
What are the advantages of Captify™ assay services?
EpiCypher has considerable experience in developing and optimizing chromatin binding assays, including those for chromatin readers, writer / eraser enzymes, antibodies, and more. Captify assay services leverage this expertise, combined with our comprehensive library of modified nucleosome and histone peptide substrates, to deliver highly sensitive and customized assays. There are multiple advantages to using Captify assay services:
- Detect chromatin interactions missed by histone peptide arrays
- Screen against physiologically relevant targets, with access to >80 modified nucleosome substrates
- Assess the binding of histone binding multiple domains in one experiment
- Reduced protein input requirements (average is nM)
- Accommodates full length proteins
- Prior knowledge of histone PTM interactions not necessary
- Have option of profiling against our library of ~300 modified histone peptides, including single and combinatorial histone PTMs
What are the service applications?
Applications for Captify assay services include:
- Drug target identification and validation
- Reveal novel biological mechanisms
- Determine how modifications impact chromatin enzyme activity
- Examine chromatin reader binding specificity
Publications and Technical Notes
The Captify platform is starting to make a significant impact in epigenetics research. Learn more about our most recent collaboration, in which the Captify approach was utilized to support the theorized mechanism of action of NuA4.
In another collaboration, the Captify platform was instrumental in revealing the mechanism of DNMT3A recruitment at intergenic regions.
EpiCypher has also published a series of Technical Notes to help scientists use the Captify platform in their chromatin research.
- AF9 YEATS Domain Acyl-Modified Histone Binding Assay: AlphaScreen Technology Tech Note
- ENL YEATS Domain Acyl-Modified Histone Binding Assay: AlphaScreen Technology Tech Note
Resources
Looking for additional information on Captify? Click through to learn more!
- Should you use histone peptides or recombinant nucleosomes to study chromatin interactions? Learn about it on our blog!
- Interested in using Captify in your lab? Shop our diverse selection of Captify Nucleosome Panels here!
- For information on the Captify platform, see our Brochure.