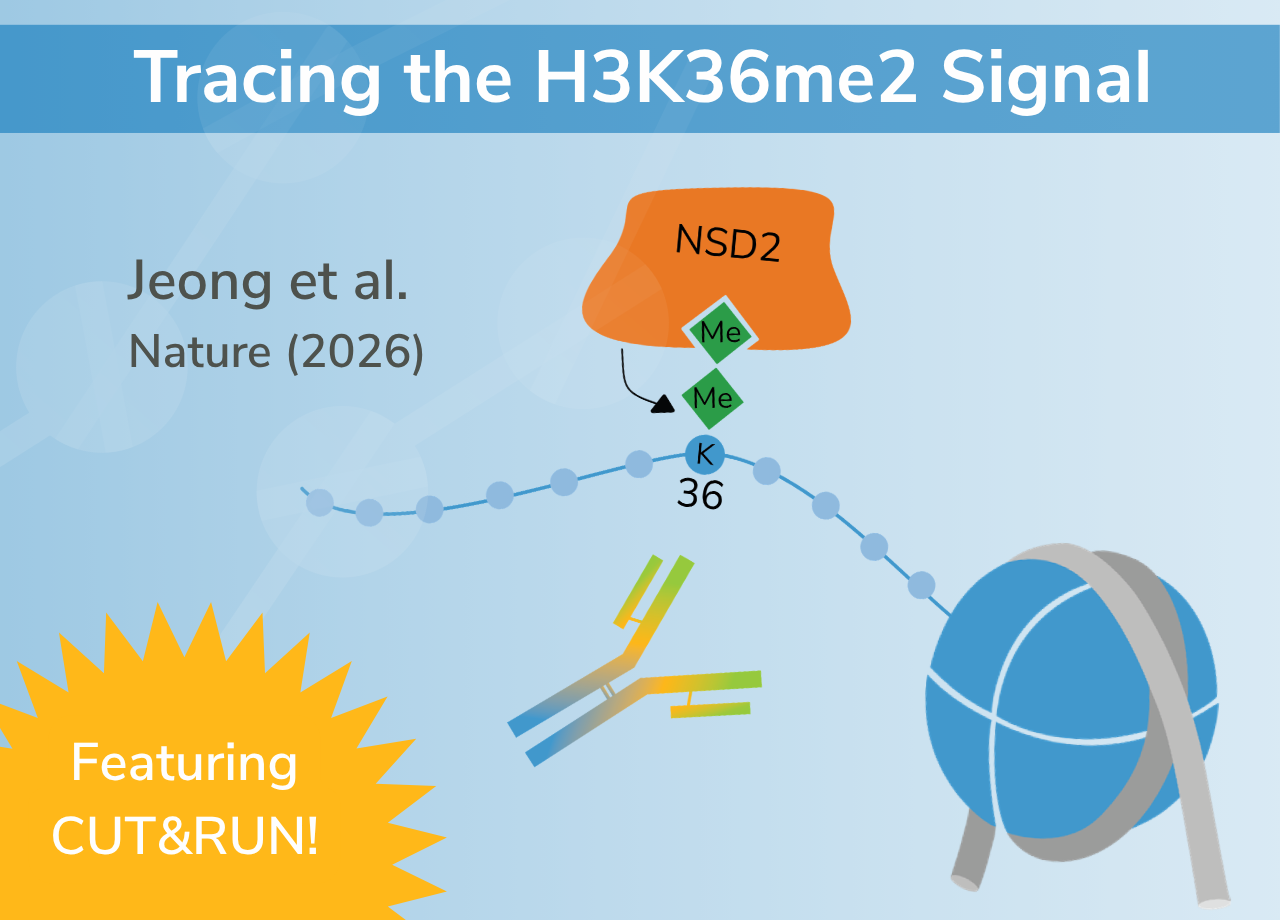
Tracing the H3K36me2 Signal: CUT&RUN Enabled Insights into NSD2 Inhibitor Response
- Tessa M. Firestone
Interrupting the epigenetic programs that drive tumor growth is a promising avenue for cancer therapy, and NSD2 is emerging as a compelling target. However, robust technologies are needed to define the mechanism of action of NSD2-targeted therapeutics, particularly their direct effects on chromatin dynamics that influence downstream gene expression changes and drug response.
NSD2 is a histone methyltransferase responsible for depositing H3K36me2, a modification broadly associated with active chromatin and transcriptional maintenance.1-3 In cancer, NSD2 is frequently altered through activating mutations, translocations, or overexpression, leading to elevated H3K36me2 and reduced levels of the repressive mark H3K27me3.3-5 This shift promotes oncogenic chromatin states and has been well characterized in multiple myeloma and increasingly in solid tumors.
At the same time, loss of NSD2 activity disrupts normal development, highlighting the importance of balanced H3K36 methylation.4 Together, these findings position NSD2 as an important regulator to target for epigenetic therapies.
Targeting NSD2 to Rewire Oncogenic Chromatin
A recent Nature study (Jeong et al. Nature, PMID: 40770093) explored whether inhibiting NSD2 could reverse oncogenic chromatin states in KRAS-driven lung and pancreatic cancers. The results point to a clear epigenetic shift, with CUT&RUN helping to define how chromatin states unfold across the genome.
NSD2 inhibition led to:
- Loss of H3K36me2
- Gain of H3K27me3
- Reprogramming of transcriptional networks linked to tumor growth
Using RNA-seq, the authors found that NSD2 inhibition downregulated more genes than it upregulated, with many of the suppressed genes linked to oncogenic pathways. Together, these data suggest that NSD2 activity helps sustain transcriptional programs required for tumor maintenance. When combined with KRAS inhibitors, NSD2 inhibition produced even stronger effects, including tumor regression and improved survival, highlighting the potential of targeting chromatin regulators alongside oncogenic signaling pathways.
Still, while RNA-seq revealed the downstream transcriptional consequences of NSD2 inhibition, a chromatin profiling approach was needed to understand the mechanism underlying those changes.
Mapping Chromatin Changes with CUT&RUN
A key strength of this study was the ability to directly measure chromatin changes across the genome.
Using CUT&RUN, researchers profiled H3K36me2 alongside additional histone marks and NSD2 itself. This approach enabled high-resolution mapping of where chromatin states changed following inhibitor treatment.
Rather than relying on bulk measurements, CUT&RUN revealed:
- Where H3K36me2 was depleted across the genome
- How these changes aligned with transcriptional outcomes
- How NSD2 activity shapes chromatin organization at specific loci
Across both cell models and pancreatic ductal adenocarcinoma (PDAC) mouse model, NSD2 inhibition reduced H3K36me2 at oncogenic gene targets. This promoted the formation of facultative heterochromatin, decreased transcriptional output, and ultimately suppressed tumor growth.
These findings position H3K36me2 as a functional readout of NSD2 activity linking chromatin state directly to gene regulation.
Enabling Insight with Validated Chromatin Profiling Tools
High-resolution chromatin profiling depends on reagent quality.
In this study, CUT&RUN experiments were performed using EpiCypher’s H3K36me2 antibody (13-0056) alongside a panel of validated histone PTM antibodies (Table 1). These tools enabled confident detection of biologically relevant signals and supported mechanistic insights into epigenomic reprogramming.
EpiCypher’s validation strategy is built to support confidence in chromatin profiling data. For histone PTM antibodies, every antibody lot is evaluated in CUT&RUN and/or CUT&Tag workflows. Antibody specificity is tested directly in the assay workflow using SNAP-CUTANA™ spike-in controls, which provide a quantitative measure of on- vs. off-target enrichment using a panel of related histone PTMs. Biological signals are assessed at multiple levels, including checking peaks at known loci, and evaluating genome-wide enrichment metrics to ensure high signal-to-noise and alignment with the expected target biology. By combining lot-specific genomic validation, in-assay specificity testing, and both locus-level plus genome-wide evaluation, EpiCypher helps ensure that antibody performance reflects true biological signal that is reliable over time.
As chromatin mapping technologies continue to evolve, pairing robust assays like CUT&RUN with well-validated reagents is essential for generating reproducible, biologically meaningful data.
| Table 1: EpiCypher reagents used in this study | |
|---|---|
Antibodies | Catalog # |
| CUTANA™ IgG Negative Control Antibody for CUT&RUN and CUT&Tag | 13-0042 |
| H3K4me1 Antibody, SNAP-Certified™ for CUT&RUN and CUT&Tag | 13-0057 |
| H3K4me3 Antibody, SNAP-Certified™ for CUT&RUN | 13-0041 |
| H3K27me3 Antibody, SNAP-Certified™ for CUT&RUN and CUT&Tag | 13-0055 |
| H3K36me2 CUTANA™ CUT&RUN and CUT&Tag Antibody | 13-0056 |
| H3K36me3 Antibody, SNAP-Certified™ for CUT&RUN | 13-0058 |
CUTANA™ ChIC/CUT&RUN Reagents | Catalog # |
| CUTANA™ Concanavalin A-Conjugated Paramagnetic Beads | 21-1401 |
| SNAP-CUTANA™ K-MetStat Panel | 19-1002 |
| CUTANA™ pAG-MNase for ChIC/CUT&RUN Workflows | 15-1016 |
| CUTANA™ Library Prep kit | 14-1001 |
| CUTANA™ ChIC / CUT&RUN Manual and DIY protocols | |
A New Lens on Epigenetic Therapeutics
This work highlights a broader shift in how researchers’ study and target epigenetic regulation in disease.
By profiling H3K36me2, scientists can:
- Track NSD2 activity across the genome
- Link chromatin changes to transcriptional outcomes
- Evaluate responses to targeted therapies
As interest in epigenetic therapeutics continues to grow, approaches that provide a direct readout of drug mechanism of action will be critical for accelerating discovery. CUT&RUN fulfills this niche, filling in the missing link between drug treatment and downstream transcriptional changes by uncovering changes in the chromatin landscape.
Apply These Insights to Your Research
Studies like this demonstrate the power of combining targeted inhibition with precise chromatin mapping.
Whether you are investigating epigenetic drivers of disease or evaluating new therapeutic strategies, CUT&RUN and high-quality histone PTM antibodies provide a reliable framework for uncovering meaningful biological insight. Discover the full range of EpiCypher’s resources including antibodies and affinity reagents, complete CUT&RUN solutions, and expert technical support to help streamline and drive chromatin research.
References
[1] Kuo et al. Mol Cell, 2011. PMID: 22099308
[2] Li et al. Cell Mol Life Sci, 2019. PMID: 31147750
[3] Chen et al. Genome Res, 2022. PMID: 35396277
[4] Azagra and Cobaleda. Int J Mol Sci, 2022 PMID: 36232375
[5] Li et al. Nature, 2026 PMID: 41299174