H3K36me2 CUTANA™ CUT&RUN and CUT&Tag Antibody
In stock
This H3K36me2 antibody is validated for chromatin profiling of histone H3 lysine 36 dimethylation (H3K36me2), an epigenetic mark deposited primarily by the histone methyltransferase NSD2 (WHSC1/MMSET). H3K36me2 is broadly enriched downstream of transcription start sites across gene bodies and intergenic chromatin [1], where it contributes to chromatin accessibility, transcriptional regulation, and enhancer activity [2]. Dysregulation of NSD2-mediated H3K36me2 deposition drives oncogenic transcriptional programs in multiple cancers, making H3K36me2 antibodies essential tools for studying NSD2 biology and epigenetic drug discovery.
Key advantages include:
- Target-specific enrichment in CUT&Tag – This antibody meets EpiCypher’s SNAP-Certified™ criteria in CUT&Tag, demonstrating <20% cross-reactivity to related histone PTMs when tested with SNAP-CUTANA™ controls (EpiCypher 19-1002), the gold standard for quantitative specificity assessment in chromatin profiling assays (Figure 1).
- Knockout tested – Antibody-generated CUT&Tag enrichment is depleted following NSD2 knockout, confirming that the observed signal reflects NSD2-dependent H3K36me2 deposition (Figure 4).
- Best-in-class performance in CUT&RUN – While SNAP CUTANA™ spike-in analysis indicates this antibody does not meet EpiCypher specificity thresholds for CUT&RUN, it delivers substantially stronger and more reproducible genomic enrichment than alternative commercial antibodies (Figures 5-7).
- Reliable lot-to-lot performance – Each production lot undergoes rigorous quality control, including direct testing in genomic mapping assays, to ensure consistent performance.
- Validated in drug discovery research – This antibody was used to profile changes following treatment with a novel NSD2 inhibitor, helping reveal the epigenomic consequences of NSD2 inhibition, demonstrating its utility for studying NSD2 biology, and evaluating epigenetic therapeutics [3].
Figure 1: SNAP specificity analysis in CUT&Tag
CUT&Tag was performed as described. CUT&Tag sequencing reads were aligned to the unique DNA barcodes corresponding to each nucleosome in the K-MetStat panel (x-axis.) Data are expressed as a percent relative to on-target recovery (H3K36me2 set to 100%). Dotted line represents the 20% cross reactivity threshold for SNAP-certification.
Figure 2: CUT&Tag genome-wide enrichment
CUT&Tag was performed as described. Sequencing reads were aligned to 23,253 annotated genes and heatmaps were generated across gene bodies from TSS to TTS extending 2kbp upstream from the TSS and 2kbp downstream from the TTS. Signal enrichment was sorted from highest to lowest (top to bottom) relative to H3K36me2 sample (all genes row aligned). High, medium, and low intensity are shown in red, yellow, and blue, respectively. H3K4me3 positive control and H3K36me2 antibodies produced the expected enrichment pattern, which were higher than those observed with the IgG negative control.
Figure 3: H3K36me2 CUT&Tag representative browser tracks
CUT&Tag was performed as described above. Gene browser shots were generated using the Integrative Genomics Viewer (IGV, Broad Institute). H3K36me2 antibody tracks display the characteristic enrichment known to be consistent with the function of this PTM [2].
Figure 4: Profiling NSD2+/- cells reveals the downstream reduction of H3K36me2
CUT&Tag profiling demonstrates that the loss of NSD2 reduces H3K36me2 enrichment across intergenic regions, reshaping the regulatory chromatin architecture. A, Immunoblot of KMS11 multiple myeloma cells that have a t(4;14) translocation resulting in NSD2 overexpression (Control; left) and isogenic clones with the translocated copy of NSD2 genetically ablated by CRISPR/Cas9 (NSD2+/-; right). B, Normalized reads counts for K-MetStat spike-ins (EpiCypher 19-1002) for CUT&Tag performed in WT and NSD2+/- KMS11 cells with H3K36me2 (EpiCypher 13-0056) and negative control IgG (EpiCypher 13-0042) antibodies. C, Gene track of H3K36me2 enrichment at the MCL1 locus in KMS11 Control and NSD2+/- cells. CUT&Tag was performed with CUTANA™ CUT&Tag Kit v1 (EpiCypher 14-1102).
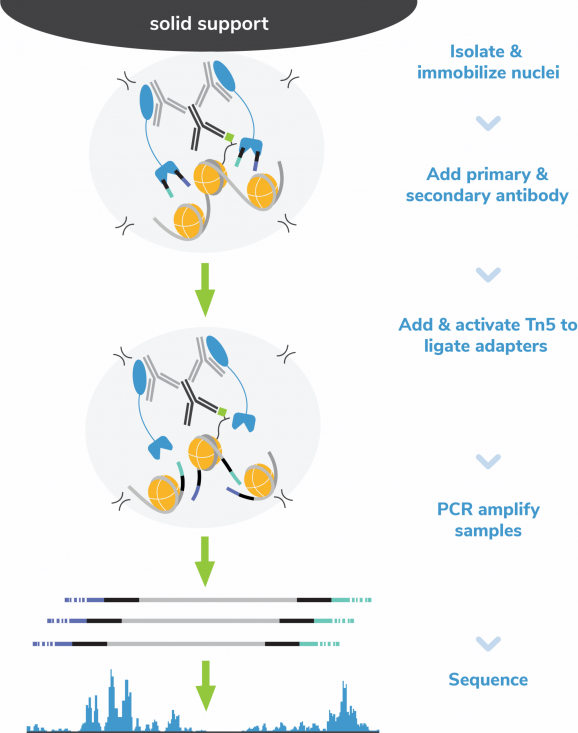
CUT&Tag methods
CUT&Tag was performed on 100k K562 nuclei with the SNAP-CUTANA™ K-MetStat Panel (EpiCypher 19-1002) spiked-in prior to the addition of 0.5 µg of either IgG negative control (EpiCypher 13-0042), H3K4me3 (EpiCypher 13-0060), or H3K36me2 antibodies. The experiment was performed using the CUTANA™ CUT&Tag Kit v4 (EpiCypher 14-1102). Libraries were run on an Illumina NextSeq2000 with paired-end sequencing (2×50 bp). Sample sequencing depth was 11.8 million reads (IgG), 10 million reads (H3K4me3), and 8.4 million reads (H3K36me2). Data were aligned to the hg38 genome using Bowtie2. Data were filtered to remove duplicates, multi-aligned reads, and ENCODE DAC Exclusion List regions.
Figure 5: SNAP specificity analysis in CUT&RUN
EpiCypher H3K36me2 antibody is best-in-class when tested against a panel of highly cited commercial antibodies. CUT&RUN was performed as described using SNAP-CUTANA™ K-MetStat spike-in controls for in-assay specificity testing (EpiCypher 19-1002). Data are expressed as a percent relative to on-target recovery with dark red indicating binding values greater than 100%. Although the EpiCypher’s H3K36me2 antibody (13-0056) cross-reacted with H3K36me1 (50%) and H3K4me2 (67%), it demonstrated the highest specificity amongst four alternate H3K36me2 antibodies (Ab1-4) that have been widely used in the literature (based on citation counts listed on individual product pages, accessed March 18, 2026; shown in purple). H3K4me3 and IgG positive and negative controls, respectively, are shown for comparison. Data shown are representative and do not represent lot-specific testing.
Figure 6: CUT&RUN genome-wide enrichment
EpiCypher’s H3K36me2 antibody produced the expected genome-wide gene body enrichment pattern. CUT&RUN signal was assessed across 23,253 annotated genes, spanning gene bodies from TSS to TTS with 2 kbp flanking regions upstream and downstream. Heatmaps were row-aligned and sorted by signal intensity relative to EpiCypher’s H3K36me2 sample. As expected, the positive control (H3K4me3) and EpiCypher’s H3K36me2 antibody showed clear enrichment above the negative control (IgG). In contrast, Three of the four highly-cited alternate antibodies (Ab1–4) showed little to no enrichment, while Ab2 produced a pattern resembling H3K4me3. Data shown are representative and do not represent lot-specific testing.
Figure 7: H3K36me2 CUT&RUN representative browser tracks
EpiCypher H3K36me2 antibody produced the expected enrichment consistent with the known distribution of H3K36me2, while highly cited alternate antibodies failed. Gene browser views, (IGV, Broad Institute), from two representative loci showed that the CUT&RUN kit generated the expected patterns for positive (H3K4me3, H3K27me3) and negative (IgG) controls. EpiCypher’s H3K36me2 antibody showed signal localization consistent with the expected genomic distribution of the target mark [2]. Whereas, the four highly cited alternate antibodies performed poorly, including Ab2, which resembled a H3K4me3 enrichment pattern. Data shown are representative and do not represent lot-specific testing.
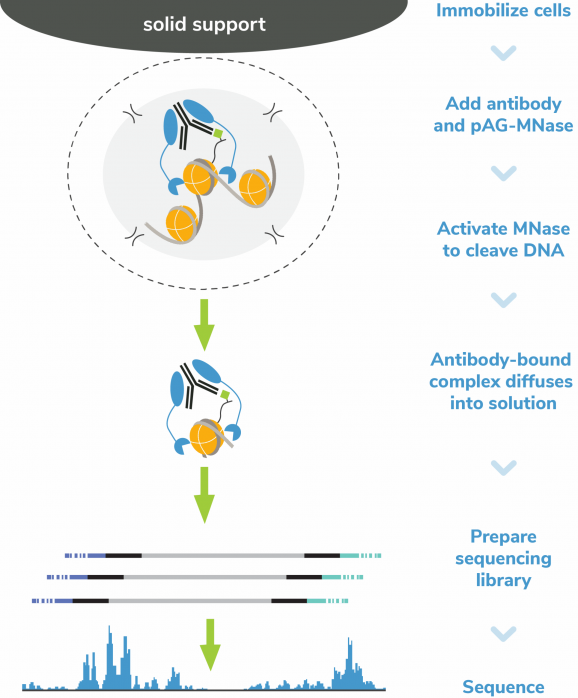
CUT&RUN methods
CUT&RUN was performed on 500k K562 cells with the SNAP-CUTANA™ K-MetStat Panel (EpiCypher 19-1002) spiked-in prior to the addition of 0.5 µg of either IgG negative control (EpiCypher 13-0042), H3K4me3 (EpiCypher 13-0060), H3K27me3 (EpiCypher 13-0055), or H3K36me2 antibodies (EpiCypher 13-0056 and five additional highly-cited commercial antibodies, referred to as Ab1-4). The experiment was performed using the CUTANA™ ChIC/CUT&RUN Kit (EpiCypher 14-1048). Libraries were run on an Illumina NextSeq2000 with paired-end sequencing (2×50 bp). Sample sequencing depth was 7.4 million reads (IgG), 9 million reads (H3K4me3), 9.8 million reads (H3K27me3), and 8.3 million reads (H3K36me2). Sequencing depth for the highly-cited antibodies was 6.1 million, 14.9 million, 38.2 million, and 5.9 million respectively. Data were aligned to the hg38 genome using Bowtie2. Data were filtered to remove duplicates, multi-aligned reads, and ENCODE DAC Exclusion List regions.
- Type: Monoclonal [2527-1B4]
- Host: Rabbit
- Applications: CUT&Tag, CUT&RUN
- Reactivity: Human, Wide Range (Predicted)
- Format: Protein A affinity-purified
- Target Size: 15 kDa
- CUT&Tag: 0.5 µg per reaction
- CUT&RUN: 0.5 µg per reaction
- UniProt ID: H3.1 – P68431
- Alternate Names: H3, H3/a, H3/b, H3/c, H3/d